1. Bis(dichlorophosphino)methane (dcpm)
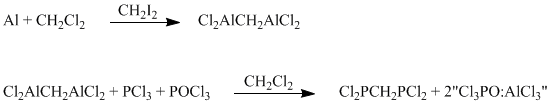
Procedure:
Caution! The aluminum and phosphorous chloride compounds are extremely water sensitive. This preparation should be carried out in a good fume hood with appropriate precautions.
2 x 1 L three-neck round bottom flasks, equipped with magnetic stirrers, magnetic stir bars, reflux condensers, and rubber septa, were charged with Al foil (16.2 g each, 0.60 mol, cut into 2 x 2 cm sections). The apparatus were evacuated and filled with nitrogen. The CH2Cl2 (~500 mL, dried over CaH2 and degassed) was added to each apparatus via a stainless steel cannula. The mixtures were heated to reflux at which time CH2I2 (~2 mL each) was added via syringe to initiate the reaction. More CH2I2 may be needed if it is noticed a reaction has not started after several hours. The mixtures were refluxed until all of the Al foil had reacted (12-24 h) to give dark amber solutions of Cl2AlCH2AlCl2 (dcam). The dcam solutions were allowed to cool to room temperature and any insoluble material was allowed to settle out. Do not allow the dcam solutions to sit for extended periods of time since dcam will precipitate out.
A 2 or 3 L three-neck round bottom flask equipped with a mechanical stirrer, reflux condenser, and septum was evacuated and filled with nitrogen. To this apparatus was added PCl3 (MCB, 104 mL, 1.2 mol, degassed) and CH2Cl2 (~300 mL, dried and degassed). The dcam solutions were slowly added via cannula over a period of 1-2 h. None of the solids produced in the dcam reactions were added. During addition, the CH2Cl2 starts to reflux from the exothermic reaction between dcam and PCl3. After addition, the mixture was refluxed 2-3 h then cooled to room temperature. The POCl3 (Fischer Scientific, 112 mL, 1.2 mol, degassed) was slowly added via cannula over a period of 1 h. This addition was also accompanied by an exothermic reaction causing the CH2Cl2 to reflux. The mixture was refluxed for 3-4 h then cooled to room temperature. Any solids present were allowed to settle out. The remaining solution was transferred to a 1 L one neck round bottom flask and concentrated on a rotary evaporator equipped with a nitrogen supply. The solid remaining in the reaction flask was thoroughly washed with ~200 mL portions of CH2Cl2. The extract solutions were added to the original solution. The solution was concentrated down to a paste. The brown paste was vacuum distilled to remove all volatiles. Distillation was continued until AlCl2, from the decomposition of Cl3PO:AlCl3, began to sublime. The distillate was redistilled under nitrogen to remove any CH2Cl2 and PCl3 present. The POCl3 was removed under reduced pressure (0.5 mm Hg) at 25°C. The Cl2PCH2PCl2 (dcpm) was distilled at 45-50°C/0.5 mm Hg to give 35-40 mL (44-50%) colorless liquid.
Caution! Care should be taken when cleaning up the residues from the dcam and dcpm reactions. Slowly react the residue first with isopropyl alcohol then with H2O behind a safety shield.
Properties:
The product exists as colorless to yellow liquid which is air sensitive and highly water sensitive. 1H NMR (CDCl3, TMS int): δ 3.51 (t, JPH = 16.4 Hz). 13C NMR (CDCl3, TMS int): δ 55.6 (t, JCP = 66 Hz). 31P{1H} NMR (CDCl3, 85% H3PO4 ext): δ 175 (s).
References:
1. Kubiak, C. P.; Kullberg, M. L.; Lemke, F. R.; RaghuVeer, K. S.; King, C.; Roundhill, D. M. Inorg. Synth. in press.
2. Novikova, Z. S.; Prishchenko, A. A.; Lutsenko, I. F. Zh. Obshchei. Khim. 1977, 47, 775.
3. Ort, M. R.; Mottus, E. H. J. Organomet. Chem. 1973, 50, 47.
2. Bis(dimethylphosphino)methane (dmpm)
![]()
Procedure:
Caution! Dmpm is extremely air sensitive and must be prepared under very oxygen free conditions. Also, dmpm is a highly toxic compound with an unbearable obnoxious odor. It is absorbed through the skin very rapidly. Inhalation or absorption of this compound, even in minute quantities, causes severe headache, nausea, blurred vision, fainting, rise in body temperature, weakness of limbs, and changes in temperament. It is imperative to handle this compound in an efficient hood and gloves must be used. All vapors from the apparatus must be passed through bromine or sodium hypochlorite solutions before release to hood exhaust.
Methyl magnesium bromide (Aldrich, 142 mL, 0.43 mol, 3 M in diethyl ether) was siphoned under nitrogen into a 3-neck flask containing diethyl ether (100 mL anhydrous, degassed) and fitted with a mechanical stirrer, a Friederichs condenser, and a pressure-equalized dropping funnel. The flask was immersed in a -10 °C) bath. The dcpm (13.4 mL, 0.10 mol) was dropped in slowly, over a period of 0.5 h, while the contents of the flask were stirred. After this addition was complete the bath was removed and the flask was slowly brought to room temperature without interrupting the stirring. The cooling bath was replaced and maintained at -10 - 0°C. A solution of ammonium chloride (47 g) in deoxygenated water (150 mL) was dropped in slowly over a 2 h period without interrupting the stirring.
The solution of ammonium chloride was added to hydrolyze the unreacted Grignard reagent and dissolve the magnesium halides. In one experiment this step was omitted and the yield of dmpm was only 55%. Since dmpm is easily oxidized, the water was thoroughly deoxygenated. In some trials, the magnesium halides did not completely dissolve and acetic acid was added (hydrochloric acid must not be used as it reacts with dmpm).
The stirring was stopped and when the contents of the flask had settled much of the aqueous layer was siphoned out. The remainder of the liquid was transferred to a separatory funnel and separated. The aqueous portion was re-extracted with two 100 mL portions of diethyl ether. Diethyl ether was removed by distillation at atmospheric pressure (diethyl ether must not be removed in a flash evaporator to save time as much of the product will be carried over with the ether). The residue was subjected to vacuum distillation. Distillate boiling at 45-50°C/10-15 mm was collected in a flask kept at -78°C. Yield: 12-13 g (88 - 96%).
Properties:
The product exists as an extremely air sensitive colorless liquid which fumes on contact with small amounts of oxygen. 1H NMR (CDCl3, TMS int): δ 1.40 (t, 2JPH = 0.7 Hz, CH2), δ 1.08 (t, N = (2JPH + 4JP'H) = 3.0 Hz, CH3). 13C{1H} NMR (CDCl3, TMS int): δ 15.8 (t, JCP = 2 Hz, CH3), δ 34.9 (t, JCP = 20 Hz, CH2). 31P{1H} NMR (CDCl3, 85% H3PO4 ext): δ -56.5 (s).
References:
1. Kubiak, C. P.; Kullberg, M. L.; Lemke, F. R.; RaghuVeer, K. S.; King, C.; Roundhill, D. M. Inorg. Synth. in press.
2. Karsch, H. H. Z. Naturforsch. 1983, 38B, 1027.
3. Fild, M.; Heinze, J.; Kruger, W. Chem Ztg. 1977, 101, 259.


