1. Preparation of Ni2(CNMe)3(dppm)2
NiCp2 + 4CNMe → Ni(CNMe)4
2Ni(CNMe)4 + 2dppm → Ni2(CNMe)3dppm)2 + 5CNMe
Procedure:
All manipulations were done under N2. To a solution of NiCp2 (1 g) in ether was added 1.6 mL of CNMe at 0�C. The reaction led to the formation of Ni(CNMe)4 as a light yellow solid. After 1 h, the solid was collected, rinsed with hexane, and dried under vacuum: Yield 80%.
To a suspension of Ni(CNMe)4 in C6H6 was added one equivalent of dppm in C6H6. The reaction solution was stirred for several hours. Part of the solvent was removed under vacuum in order to remove the liberated CNMe. More C6H6 was added and stirring continued. This procedure was repeated at least three times before the solution was concentrated and hexanes added to initiate precipitation. The solution was cooled to -20�C overnight. Ni2(CNMe)3(dppm)2 was isolated and washed with hexane, then MeCN to remove any impurities. Yield 60-70%.
Properties:
Formula weight: 1008.3 g/mol; 1H NMR (C7D8, -30 �C) d 2.44 (s, 3H), 2.55 (s, 6H), 4.31 (m, 4H), 7.2-8.1 (m, 40H); 31P{1H} NMR (C7D8, 25 �C) d 18.21 (s); IR (toluene) �(CN) 2075, 1717 cm-1.
References:
1. DeLaet, D. L.; Powell, D. R.; Kubiak, C. P. Organometallics 1985, 4, 954.
2. DeLaet, D. L.; Fanwick, P. E.; Kubiak, C. P. Organometallics 1986, 5, 1807.
3. Nickelocene is commercially available from Aldrich or can be prepared according to the literature preparation: Barnett, K. W. J. Chem. Educ. 1977, 51 422.
2. Preparation of Ni2(CO)3(dppm)2
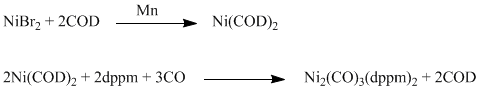
Procedure:
Anhydrous NiBr2, 24 g, was dissolved in 150 mL of ethylene glycol monomethyl ether(methyl cellosolve) by refluxing. The solution was cooled and 50 mL of 1,5-cyclooctadiene and 20 mL of quinoline added. The mixture was cooled to -10�C and 8 g of finely powdered manganese was added under nitrogen with stirring. After a short induction period, the solution turned orange-red (an intermediate complex with quinoline is formed) and after 15-20 min dicyclooctadienenickel(O) began to precipitate. The reaction was continued for about 1 h with stirring, the temperature of the bath being kept between -5 and -10�C. The mixture was filtered under nitrogen then washed first with cold (0�C) methyl cellosolve until the filtrate remained colorless (100-150 mL of solvent), and then with ether cooled at -30�C. The bright yellow solid obtained was dried and consisted of a mixture of dicyclooctadienenickel(O) and residual metallic manganese.
To remove the metal, dicyclooctadienenickel(0) may be crystallized as follows. The crude product is placed on a Buchner funnel under nitrogen and washed with a toluene solution (250 mL) containing 1-2 mL of quinoline and 5-10 mL of cyclooctadiene, kept at 90�C and added in small amounts (80 mL) to the Buchner funnel without sucking; the precipitate was agitated with a glass rod and filtration then carried out by applying a gentle nitrogen pressure from the nitrogen line. The process was repeated until the filtrate remained colorless. The combined filtrates were placed in a flask under nitrogen and immersed in a bath at -70�C. The dicyclooctadienenickel(O) formed was filtered, extensively washed with cold ether (at -30�C) and dried under vacuum. Yield 50-80%.
Freshly prepared Ni(COD)2 was dissolved in THF to which was added 1.1 eq dppm in a small amount of THF. The mixture is stirred for about 20 min and the color of the solution changes from yellow to orange yellow. With the flask under a slight vacuum, 1.5 - 1.6 eq. CO gas is added via syringe. The mixture is stirred for 30 min and stored at -20�C overnight.(a) A small amount of yellow precipitate forms overnight. Warm the reaction solution to 45�C and stir 30 min. Allow the reaction solution to cool to room temperature. The reaction volatiles were removed via vacuum (b) until a large amount of yellow precipitate with a small amount of solvent is left. The yellow precipitate is filtered, washed with ether and dried under vacuum: Yield 80%.
Properties:
Formula weight 970.22 g/mol; IR [KBr] �(CO) 1970, 1948, 1781 cm-1; 31P{1H} NMR [CH2Cl2] d 22.92 ppm; 1H NMR [CD2Cl2] d 2.61 (m, 2H), 3.42 (m,2H0, 6.92-7.38 (m, 40H).Notes:
(a) During the preparation, there is a possibility of forming a trace amount of Ni(CO)4. The reaction solution is stored in the freezer to prevent any Ni(CO)4 from escaping. Ni(CO)4 is a colorless, highly volatile, highly toxic liquid [b.p. 42.2 �C (760 mm Hg), IR (neat) �(CO) 2022-2038 cm-1].
(b) The reaction volatiles are collected in a liquid N2 cooled trap between the reaction flask and vacuum line. Any Ni(CO)4 is disposed of by treatment of the trap distillate with bromine water until the solution turns orange.
References:
1. Franco, G.; Giuseppe, S. J. Organomet. Chem. 1


